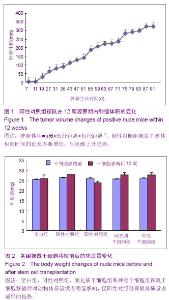
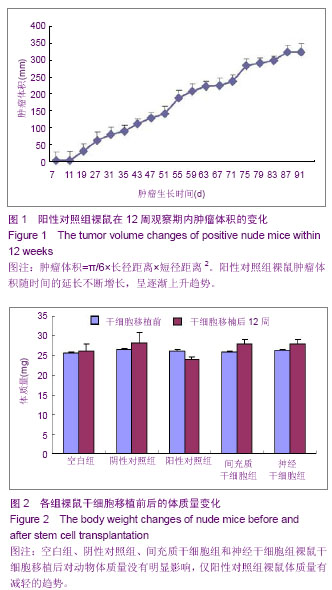
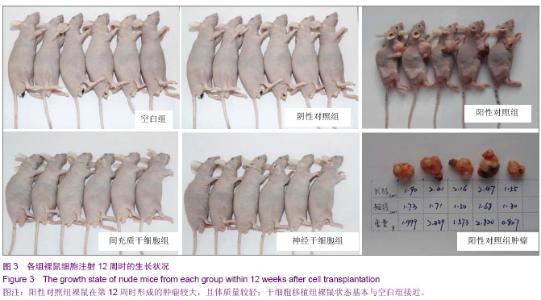
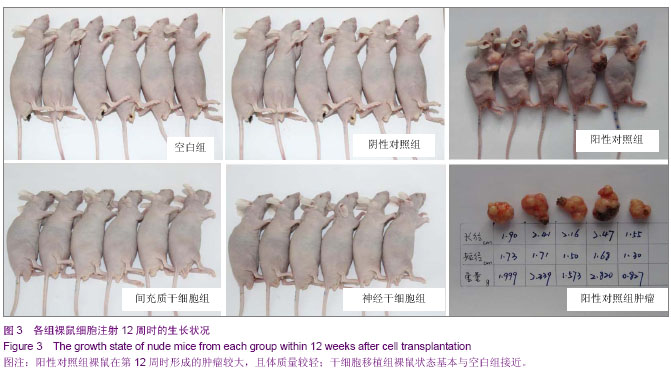
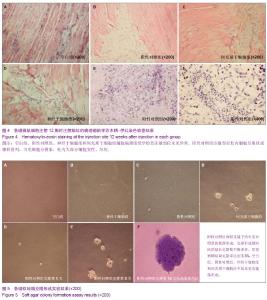
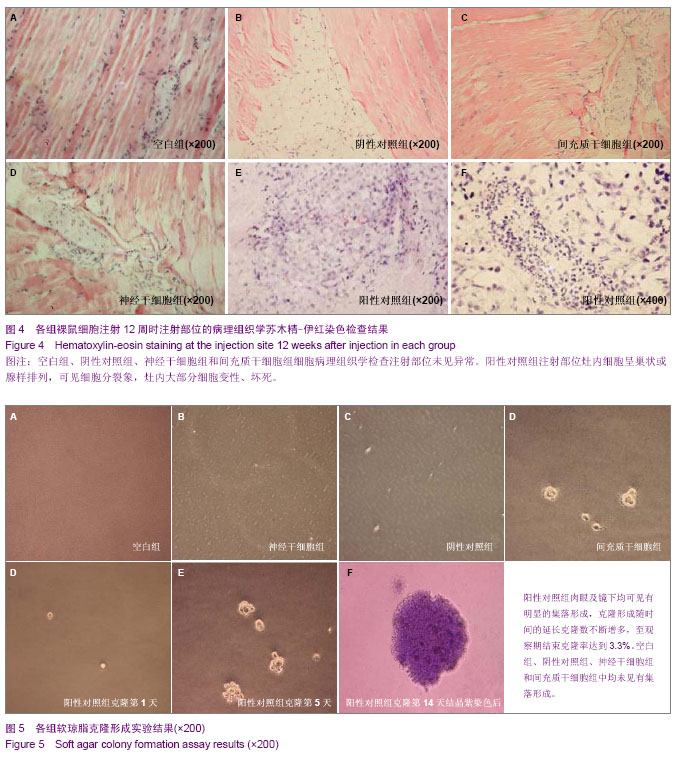
| [1] Fernandez Tde S, de Souza Fernandez C, Mencalha AL. Human induced pluripotent stem cells from basic research to potential clinical applications in cancer. Biomed Res Int. 2013; 2013:430290.[2] Greco SJ, Rameshwar P. Mesenchymal stem cells in drug/gene delivery: implications for cell therapy. Ther Deliv. 2012;3(8):997-1004. [3] Gu E, Chen WY, Gu J, et al. Molecular imaging of stem cells: tracking survival, biodistribution, tumorigenicity, and immunogenicity. Theranostics. 2012; 2(4):335-345.[4] Djouad F, Bouffi C, Ghannam S, et al. Mesenchymal stem cells: innovative therapeutic tools for rheumatic diseases. Nat Rev Rheumatol. 2009;5:392-399.[5] Teoh HK, Cheong SK. Induced pluripotent stem cells in research and therapy, Malays J Pathol. 2012;34(1):1-13.[6] Carroll SH, Ravid K. Differentiation of mesenchymal stem cells to osteoblasts and chondrocytes: a focus on adenosine receptors. Expert Rev Mol Med. 2013;15:e1.[7] Sensebé L, Fleury-Cappellesso S. Biodistribution of Mesenchymal Stem/Stromal Cells in a Preclinical Setting. Stem Cells Int. 2013;2013:678063. [8] Dorronsoro A, Fernández-Rueda J, Fechter K, et al.Human Mesenchymal Stromal Cell-Mediated Immunoregulation: Mechanisms of Action and Clinical Applications. Bone Marrow Res. 2013;2013:203643. [9] Wei X, Yang X, Han ZP, et al.Mesenchymal stem cells: a new trend for cell therapy. Acta Pharmacol Sin. 2013;34(6):747-754. [10] Stochaj U, Kodiha M, Shum-Tim D, et al. Implications of multipotent mesenchymal stromal cell aging. Regen Med. 2013;8(2):211-222.[11] Yao J, Mu Y, Gage FH. Neural stem cells: mechanisms and modeling. Protein Cell. 2012;3(4):251-261.[12] Kohman RA, Rhodes JS. Neurogenesis, inflammation and behavior. Brain Behav Immun. 2013;27(1):22-32.[13] Gincberg G, Arien-Zakay H, Lazarovici P, et al. Neural stem cells: therapeutic potential for neurodegenerative diseases. Br Med Bull. 2012;104:7-19.[14] Tan J, Meng Y, Huang S, et al.Therapeutic gene products delivery by neuron stem cells. Curr Pharm Biotechnol. 2012; 13(12):2427-2431.[15] Andressen C. Neural stem cells: from neurobiology to clinical applications. Curr Pharm Biotechnol.2013;14(1):20-28.[16] Ding DC, Lin CH, Shyu WC, et al. Neural stem cells and stroke. Cell Transplant. 2013;22(4):619-630. [17] English D, Sharma NK, Sharma K, et al. Neural stem cells-trends and advances. J Cell Biochem. 2013;114(4): 764-772. [18] Neirinckx V, Coste C, Rogister B, et al. Concise review: adult mesenchymal stem cells, adult neural crest stem cells, and therapy of neurological pathologies: a state of play. Stem Cells Transl Med. 2013; 2(4):284-296.[19] WHO Technical Report Series[R]. Annex 1: Requirements for the used of animal cells as in vitro substrated for the production of biologicals. 1998: 878. [20] FDA. Points toconsider in the characterization of cell linesused to produce biologicals[S]. 1993.[21] 中国药典.三部[S].北京: 化工业出版社, 2010: 14-17.[22] 张赛,刘振林,刘晓智,等.hTERT永生化脐带间充质干细胞的致瘤性研究[J].中国神经精神疾病, 2008, 34(3): 171-173.[23] 林戈,卢光琇.干细胞概述[J].生命科学,2006,18(4): 313-317. [24] 李凌松,路艳艳.神经干细胞及帕金森病的细胞治疗[J].北京大学学报(医学版),2002,34(5):499-505.[25] 屈红林,彭瑞. 神经干细胞研究现状及在运动医学领域中的应用[J].中国组织工程研究与临床康复,2008, 12(25):4941-4944.[26] 庞永刚,崔彭城,陈文弦,等.人骨髓间质干细胞作为骨、软骨组织工程种子细胞的实验研究[J].细胞与分子免疫学杂志,2004, 20 (3): 306-309.[27] Aggarwal S, Pittenger MF. Human mesenchymal stem cells modulate allogeneic immune cell responses. Blood. 2005; 105(4): 1815-1822.[28] Jiang WH, Ma AQ, Zhang YM, et al. Migration of intravenously grafted mesenchymal stem cells to injured heart in rats. Acta Physiological Sinica. 2005;57(5):566-572.[29] Zhu D, Wan X, Huang H, et al. Knockdown of Bmi1 inhibits the stemness properties and tumorigenicity of human bladder cancer stem cell-like side population cells. Oncol Rep. 2014; 31(2):727-736. [30] Sato Y. In vitro tumorigenicity tests for process control of health care products derived from human induced pluripotent stem cells. Yakugaku Zasshi. 2013;133(12):1381-1388.[31] de Almeida PE, Ransohoff JD, Nahid A, et al. Immunogenicity of pluripotent stem cells and their derivatives. Circ Res. 2013; 112(3):549-561.[32] Kuroda T, Yasuda S, Sato Y. Tumorigenicity studies for human pluripotent stem cell-derived products. Biol Pharm Bull. 2013;36(2):189-192. [33] Kobayashi Y, Okada Y, Itakura G, et al. Pre-evaluated safe human iPSC-derived neural stem cells promote functional recovery after spinal cord injury in common marmoset without tumorigenicity. PLoS One. 2012; 7(12): e52787.[34] Ben-David U, Benvenisty N. The tumorigenicity of human embryonic and induced pluripotent stem cells. Nat Rev Cancer.2011;11:268-277.[35] Kooreman NG, Wu JC. Tumorigenicity of pluripotent stem cells: biological insights from molecular imaging. J R Soc Interface. 2010;7(Suppl 6):S753-763. [36] Zhao T, Zhang ZN, Rong Z, et al. Immunogenicity of induced pluripotent stem cells. Nature.2011;474:212-215.[37] Shen G, Cong XQ. The tumorigenicity of immortalized cells differentiated from mouse embryonic stem cells. Zhonghua Zheng Xing Wai Ke Za Zhi. 2012;28(1):33-39.[38] Okada Y, Okano H. Evaluation of human iPS cells by neural differentiation and tumorigenicity. Rinsho Shinkeigaku. 2011; 51(11):1079-1080. [39] Dressel R. Effects of histocompatibility and host immune responses on the tumorigenicity of pluripotent stem cells. Semin Immunopathol. 2011;33(6):573-591.[40] Kleinsmith LJ, Pierce GB Jr. Multipotentiality of single embryonal carcinoma cells. Cancer Res. 1964;24:1544-1551.[41] Shamblott MJ, Axelman J, Wang SP, et al. Derivation of pluripotent stem cells from cultured human primordial germ cells. Proc Natl Acad Sci USA. 1998,95(23):13726-13731.[42] Hua JL, Qiu BP, Zhu HJ, et al. Multipotent mesenchymal stem cells (MSCs) from human umbilical cord: Potential differentiation of germ cells. Afric J of Biochem Res. 2011;5(4): 113-123.[43] 李六金,李秦,施新猷,等.五种动物传代细胞系对裸鼠的致瘤性[J]. 第四军医大学学报, 2000, 21(6):738-740. |